What Is Regulatory Dossier and What Does It Contain? - The Kolabtree Blog

Nare Simonyan - Freelance Regulatory Writing Consultant in Yerevan, Armenia

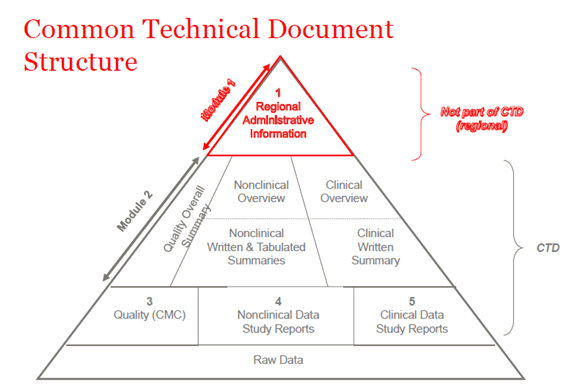
Drug development & compilation of registration dossier

About the Author

About the Author

Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Pharmaceutical Regulatory Services, CMC Writing

Medical Devices Clinical Studies / Trials Best Practices
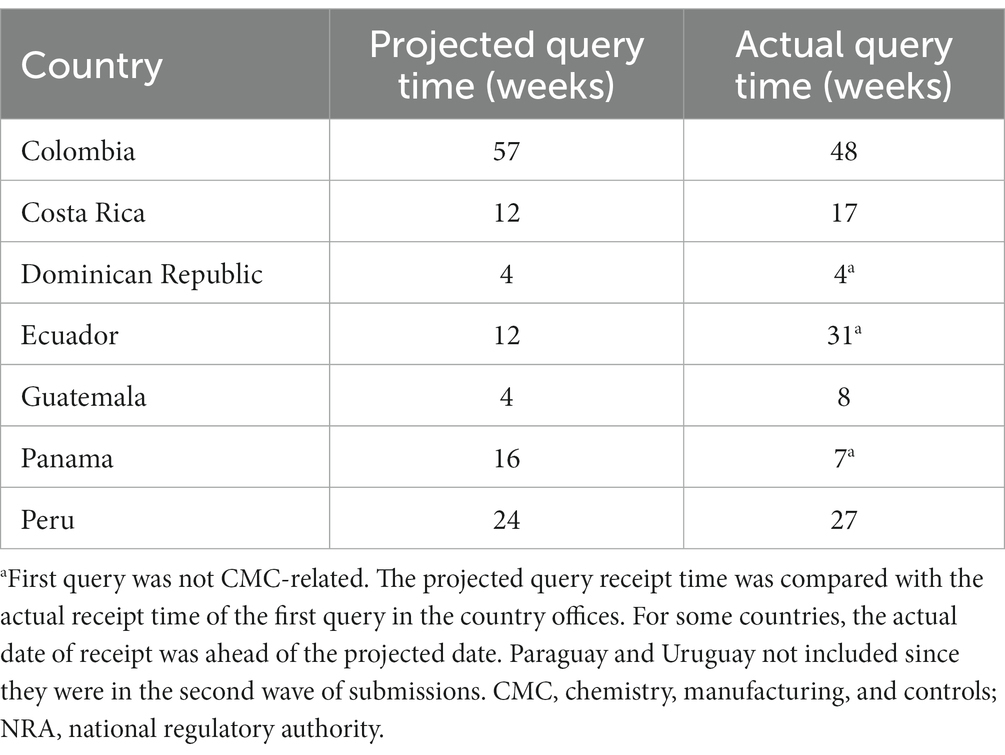
Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Exemple de rapport d'évaluation clinique d'un dispositif médical (gratuit)
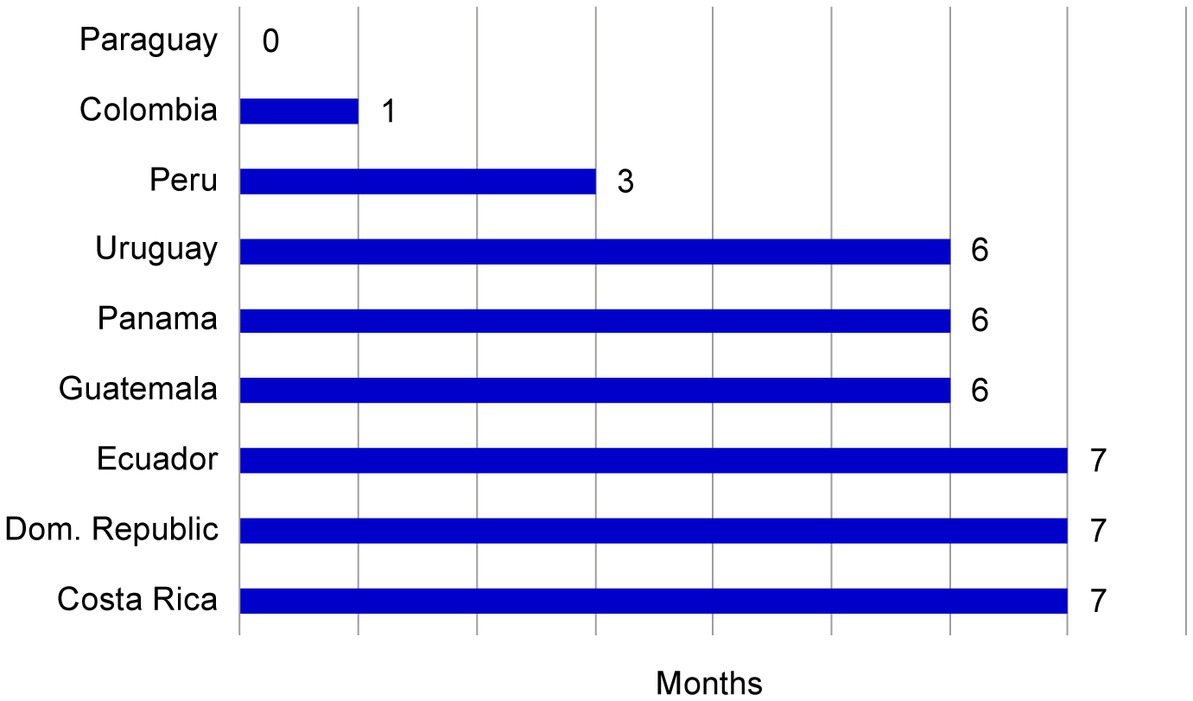
Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Notification préalable à la mise sur le marché 510k de la FDA : Exigences essentielles








