
The Submission Dossier Regulatory Affairs in Latin America
The submission dossier is the packet of documents that are to be submitted to a health authority for registration of a product, or for other life-cycle maintenance activities, such as renewal of registration or CMC variations. The requirements vary very widely from country to country, but in general a dossier contains administrative documents, (such as…

Infograph: Pharma Patents Regulatory Affairs in Latin America

Plan for self-sufficiency in health matters in Latin America and the Caribbean by Publicaciones de la CEPAL, Naciones Unidas - Issuu

Registration and post-approval variation of pharmaceutical drugs in Latin America: challenges and opportunities, Journal

eCTDmanager - Complete Regulatory Dossier Management and eCTD Software

Vault Submissions Archive, Published Output Repository

Regulatory Harmonization: Driving Forces - BioProcess InternationalBioProcess International

Registration and post-approval variation of pharmaceutical drugs in Latin America: challenges and opportunities, Journal
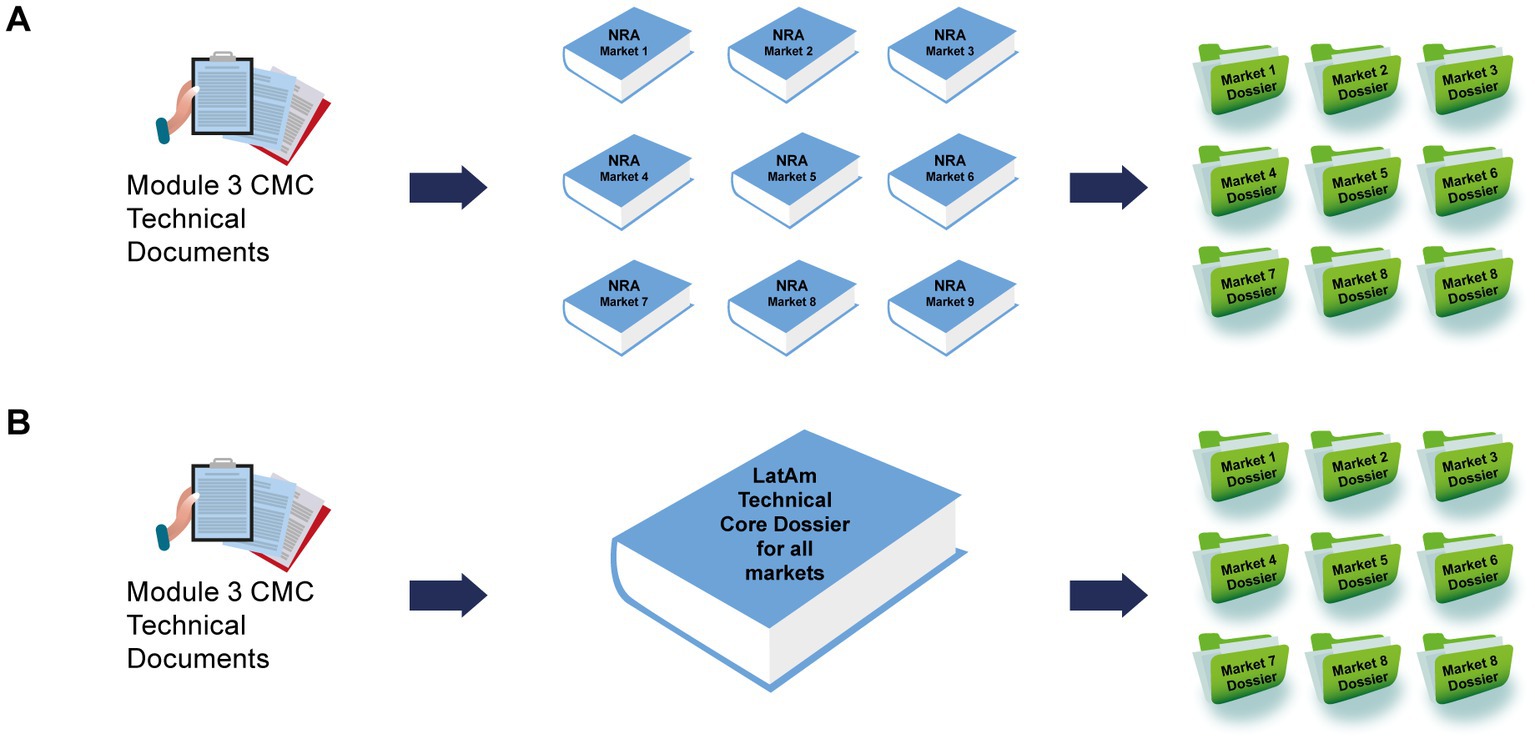
Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Regulatory Affairs Recruitment









