Performance Qualification (PQ) Definition
4.6
(526)
Écrire un avis
Plus
€ 16.50
En Stock
Description

Operational Qualification (OQ) in Pharmaceuticals : Pharmaguideline
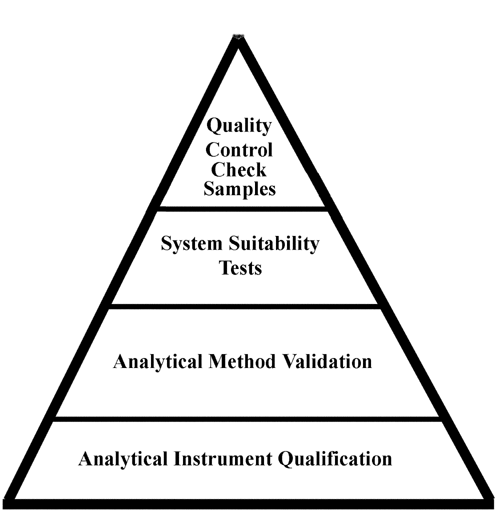
usp31nf26s1_c1058, General Chapters: <1058> Analytical Instrument

Performance Qualification (PQ) for EO Sterilization Validation

Validation Protocols - Reports Procedure
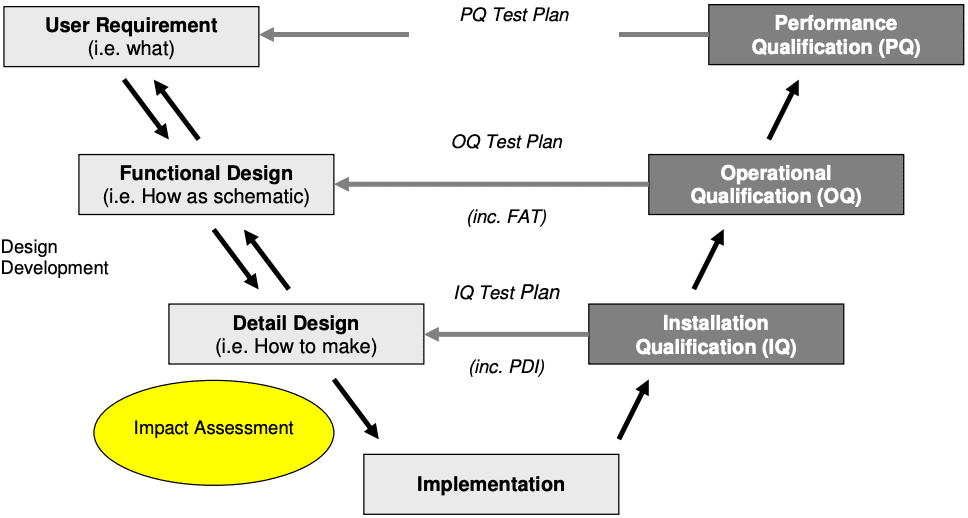
What are IQ OQ PQ? Why are they critical to the Pharma Industry?

Performance Qualification (PQ) Checklist.

VMP DQ IQ OQ PQ Validation - GMP

Why Qualification? - DQ, IQ, OQ, PQ - MicroSolv Technology Corp

What Are IQ OQ PQ, The 3 Q's Of Software Validation Process

Data Integrity and USP : Part 3: Monitoring and Requalification
Tu pourrais aussi aimer